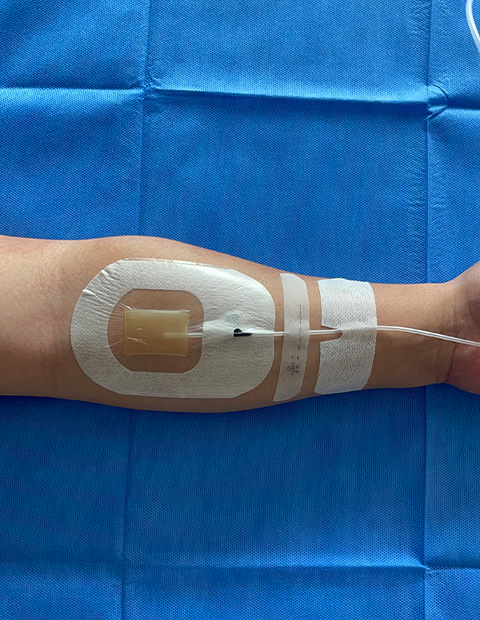
📊 Basic Information
Model NO.:CHG IV Dressing
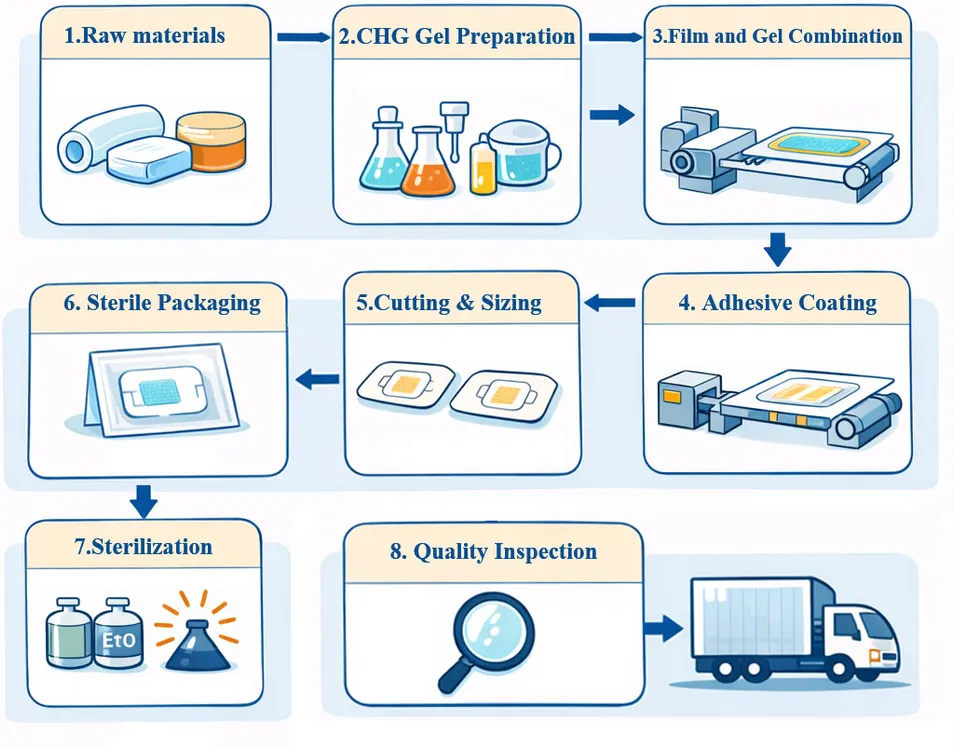
Sterilization:Ethylene Oxide (EO)
Quality Guarantee:3 Years
Advantage:CHG Antimicrobial
Application:Wound Fixation
Function:Protect Wound
Color:White Transparent
Feature:Waterproof, Latex-Free
Irritating:Non-Irritating
Certificate:CE / ISO13485
Transparency:High Transparency
Keyword:Chlorhexidine
❓ Frequently Asked Questions
Q1: Are you a manufacturer or trading company?
A: We are an integrated industry and trade company specializing in medical consumables for over 25 years.
Q2: Can I get samples for quality testing?
A: Yes, we are pleased to provide samples for your evaluation and quality testing.
Q3: Do you offer customized OEM/ODM services?
A: Yes, we support full customization for both products and packaging logos to meet your specific needs.
Q4: What is the typical lead time for orders?
A: Our standard lead time is usually 15-20 days, depending on the order size and requirements.
Q5: What certifications do your products hold?
A: Our products are fully certified with CE and ISO standards to ensure high quality and safety.
Q6: Do you have a quality inspection procedure?
A: Yes, we perform 100% self-inspection on all products before final packaging to maintain strict quality control.